
With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. 
Sulfur and oxygen both belong to the same group in the periodic table ( the chalcogen family) and have six valence electrons each. For sulfate ions, we have one molecule of sulfur and four molecules of oxygen. It is due to the resonance present in the phosphate PO 4 3- ion that each P-O bond length is equivalent as opposed to a shorter PO bond and three longer P-O bonds, as expected. Electron Domain And Molecular Geometry In chemistry and physics, the molecular geometry describes the geometric arrangement of the atoms that form a molecule. Take a look at the VSEPR chart to understand the molecular and electron geometry of BrO 2. Since the Bromine central atom has 4 regions of electron density(2 lone pairs + 2 bonded atoms), it implies, its electron domain geometry will be tetrahedral. Each O-P-O bond angle is 109.5° in PO 4 3-. The electron geometry for BrO 2 will be tetrahedral. 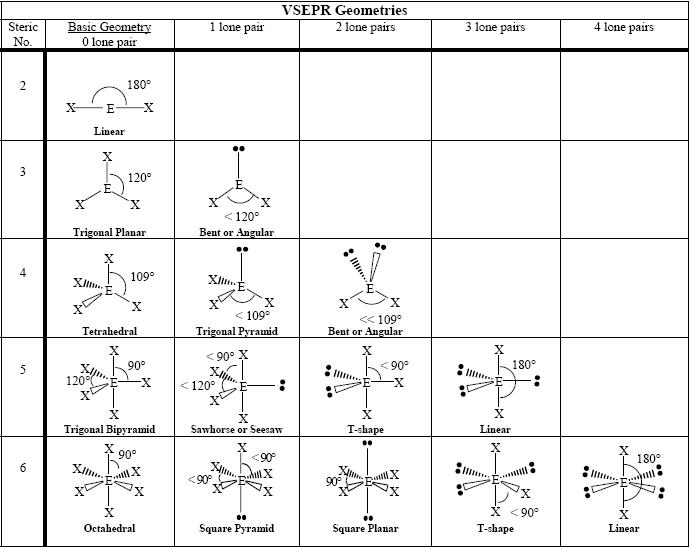
If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. Step 1: Count the total number of valence electrons present in the molecule/ion. The PO 4 3- ion has an identical electron geometry and molecular geometry or shape i.e., tetrahedral. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. Molecular geometry - The study of the three-dimensional arrangement of the atoms that constitute a molecule is called Molecular geometry. Electron Domains Electron-Domain Geometry Predicted Bond Angle(s) Hybridization of Central Atom Molecular Geometry 0 Lone Pair 1 Lone Pair 2 Lone Pair 2 Linear 180º sp Linear 3 Trigonal Planar 120º sp2 Trigonal Planar Bent 4 Tetrahedral 109. We designate SF 4 as AX 4E it has a total of five electron pairs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
